6-Methyl-5-hepten-2-one
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 110-93-0
- EC NO : 203-816-7
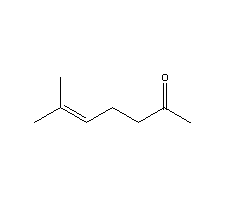
- Molecular Formula : C8H14O
- Main Specifications : 99% min
- Synonyms : 6-methylhept-5-en-one;Methyl isohexenyl ketone;
Package: 25kg��,50kg 200kg
Uses : used in spice industry and Organic synthesis
Molecular Structure:
Product description:
What is the chemical of 6-Methyl-5-hepten-2-one ?
Appearance: Light yellow transparent liquid;
Odor: It has a fruity and fresh aroma, similar to bananas, pears, citrus or lemongrass, and also similar to pentyl acetate or isobutyl acetate.
Solubility: Insoluble in water, but miscible with organic solvents such as ethanol and ether ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 172-174 ° C (at atmospheric pressure of 760 mmHg);
===============================================================================
main purpose
Spice industry: According to the Chinese national standard GB 2760-86, it is allowed to be used as food flavor, mainly used to prepare banana, pear, citrus and berry essence, but usually not directly used as flavor, but as an intermediate of synthetic flavor.
Organic synthesis: It is an important intermediate for the synthesis of medicine, essence and fragrance, and can be used to synthesize pseudoionone, citral and other compounds.
Other: Naturally occurring in guava, tomato, ginger, and tobacco, it is also an endogenous metabolite
===============================================================================
synthetic route
The detailed synthesis route is not explicitly provided in the public information, but based on its structure (including vinyl bonds and ketone groups), it can usually be synthesized through the following routes in industry:
Condensation reaction of isoprene and acetone: Under acidic or alkaline conditions, isoprene undergoes aldol condensation or similar reaction with acetone to produce 6-methyl-5-hepten-2-one.
Extracted or biosynthesized from natural sources: As it naturally exists in fruits and plants, it can also be obtained through enzymatic or fermentation methods.
