Phenethyl alcohol
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 60-12-8
- EC NO : 200-456-2
- Molecular Formula : C8H10O
- Main Specifications : 99% min
- Synonyms : 1-Phenyl-2-Ethanol;2-Phenylethanol;Alcohol Feniletilico; Benzeneethanol;Benzyl carbinol;Beta-p.e.a.;beta-Phenyl ethyl alcohol;Hydroxyethylbenzene;Mellol;Phenylethyl alcohol;2-Phenylethyl alcohol;2-Phenethyl alcohol;Phenyl ethyl alcohol;Ethanol, phenyl-;Phenylethanol;1-phenylethanol;
Package: 25kg��,50kg 200kg
Uses : used to prepare essence for soap and cosmetics
Molecular Structure:
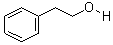
Product description:
What is the chemical of Phenethyl alcohol?
Appearance: Phenylethanol is a colorless and transparent liquid with a rose like aroma ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: -27 °C;
boiling point: 219-221 ° C (at 750 mm Hg pressure);
===============================================================================
Purpose:
It is widely used to prepare essence for soap and cosmetics, and is the main raw material for preparing rose flavor essence.
It is an edible spice allowed by Chinese regulations, which can be used for candy, baked goods, cold drinks, etc., with the dosage according to normal production needs.
Can be used as an antibacterial preservative, such as in eye drops solution.
Used as analytical standards in the field of scientific research
=============================================================================
Synthesis route:
Natural extraction: found in aromatic oils such as ylang ylang oil, orange blossom oil, and rose oil, but most commercially available products are synthetic.
Industrial synthesis method:
Styrene route: Styrene is obtained by halogenation, saponification, hydrogenation, and distillation.
Microbial fermentation method: using yeast for biotransformation.
Made from calcium carbide and benzene as raw materials: by generating styrene and then reacting with water.
