Paraldehyde
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 123-63-7
- EC NO : 204-639-8
- Molecular Formula : C6H6O2
- Main Specifications : 99% min
- Synonyms : 2,4,6-trimethyl-1,3,5-Trioxane;2,4,6-trimethyl-s-trioxane;paraacetaldehyde;Paracetaldehyde;PARAL;acetaldehyde, trimer;elaldehyde;PCHO;s-trimethyltrioxymethylene;trimethyl 1,3,5-trioxane;2H-pyran-6-carbaldehyde;
Package: 25kg�,50kg 200kg
Uses : As a stable form of acetaldehyde, trimeric acetaldehyde
Molecular Structure:
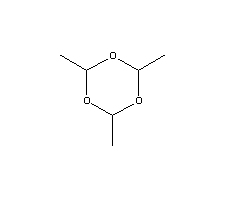
Product description:
What is the chemical of Paraldehyde?
Appearance: Colorless to pale yellow transparent liquid with aromatic odor ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 124 °C;
=============================================================================
Usage: Acetaldehyde has a wide range of uses, including:
As a stable form of acetaldehyde, trimeric acetaldehyde is easily decomposed into acetaldehyde and is therefore commonly used for safe storage and transportation of acetaldehyde. twenty-three
Solvent: widely used as a solvent and diluent for pesticides, fragrances, pharmaceuticals, and coatings.
Organic synthesis: used in the pharmaceutical industry, organic synthesis, manufacturing of rubber accelerators and antioxidants.
Other applications: In the agricultural sector, about 50% of formaldehyde is used in pesticide production in Japan, followed by the spice, pharmaceutical, and coating industries.
===============================================================================
Regarding its synthesis route, public information shows that there are multiple preparation methods, among which one efficient route is as follows:
Main synthesis route: Acetaldehyde undergoes trimerization reaction under acidic conditions (such as with a small amount of sulfuric acid catalysis) to produce trimeric acetaldehyde. This reaction is usually carried out at low temperatures to control the degree of polymerization and avoid the formation of by-products such as tetramers. The literature reports that the synthesis efficiency of this route can reach about 98%.
Other possible routes: In addition to direct acid catalyzed trimerization, it may also be achieved through multi-step reactions such as protection, activation, and cyclization of acetaldehyde, but the most commonly used method in industry and laboratory is the one-step catalytic polymerization method mentioned above.
