N,N-Diethylethylenediamine
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 100-36-7
- EC NO : 202-844-7
- Molecular Formula : C6H18N2
- Main Specifications : 99% min
- Synonyms : 2-(Diethylamino)ethylamine;2-aminoethyldiethylamine;N,N-diethylethane-1,2-diamine;N,N-diethylethane-1,2-diaminium;
Package: 25kg,50kg 200kg
Uses : as an intermediate for fine chemicals and involved in the synthesis of various drugs
Molecular Structure:
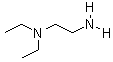
Product description:
What is the chemical of N,N-Diethylethylenediamine ?
Appearance: Colorless liquid to light yellow ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 145-147 °C;
============================================================================
Purpose:
Used as an intermediate for fine chemicals and involved in the synthesis of various drugs, such as the antiarrhythmic drug procainamide, local anesthetic bupivacaine hydrochloride, and enzyme-linked immunosorbent assay.
Used for producing auxiliary products, such as preparing Sapamine BCH.
In the field of medicine, it is also used to synthesize compounds such as procaine hydrochloride, and sulpiride hydrochloride
=======================================================================
There are several methods for synthesizing routes, including:
Prepared by hydrogenation of ethylenediaminetetranitrile: This is a common industrial synthesis pathway that produces N, N-diethylethylenediamine through catalytic hydrogenation of ethylenediaminetetranitrile.
Obtained from acrylonitrile through hydrolysis, addition, and degradation: This method involves multiple reactions. Firstly, acrylonitrile is used as the starting material, followed by intermediate steps such as hydrolysis and addition, and finally the target product is obtained through degradation reaction.
Other synthetic routes: Various possible synthetic routes have been documented in literature, such as the reaction of ethylenediamine with ethanol under specific conditions, or the alkylation reaction of other amine compounds for preparation
