Linalool
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 78-70-6
- EC NO : 201-134-4
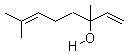
- Molecular Formula : C10H18O
- Main Specifications : 99% min
- Synonyms : (+-)-Linalool;(±)-Linalool;.beta.-Linalool;1,2-Dihydrolinalool;1,6-octadien-3-ol, 3,7-dimethyl-;1,6-Octadien-3-ol, 3,7-dimethyl-, (-)-;2,6-Dimethyl-2,7-octadien-6-ol;2,6-Dimethyl-2,7-octadiene-6-ol;2,6-dimethylocta-2,7-dien-6-ol;2,7-Octadien-6-ol, 2,6-dimethyl-;3,7-dimethylocta-1,6-dien-3-ol;
Package: 25kg,50kg 200kg
Uses : an important spice compound
Molecular Structure:
Product description:
What is the chemical of Linalool?
Appearance: colorless oily liquid with a sweet, tender, and fresh floral aroma similar to that of lily of the valley or citrus flowers;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 194-199 ° C (at 720 mmHg pressure);
=============================================================================
Usage: Linalool is an important spice compound, mainly used for:
Perfume, cosmetics and soaps: as the core component of floral essence, they are widely used to blend lily, clove, orange flower, rose and other scents, as well as amber, oriental and aldehyde essence.
Food industry: used as an edible spice to blend flavors such as lemon, orange, grape, apricot, pineapple, chocolate, etc.
Detergents and household products: used to give cleaning products a pleasant fragrance.
Pharmaceutical research: As a pharmaceutical intermediate, it has potential biological activities such as anti-tumor and anti cardiotoxicity, but its main use is still concentrated in the field of fragrances
=============================================================================
Synthesis route: There are two main ways to produce linalool:
Natural extraction: obtained by fractional distillation from natural essential oils such as rosewood oil, rosewood oil, coriander seed oil, and camphor oil.
Chemical synthesis: Using α - pinene or β - pinene from turpentine as raw materials, it is thermally decomposed to produce laurene, which then reacts with hydrogen chloride to produce linagli chloride, and finally reacts with alkali (such as caustic potassium) to produce linalool.
