Isoquinoline
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 119-65-3
- EC NO : 204-341-8
- Molecular Formula : C9H7N
- Main Specifications : 99% min
- Synonyms : 2-Azanaphthalene;2-Benzazine;3,4-Benzopyridine;AI3-10035;Benzo(c)pyridine;CCRIS 5752;FEMA No. 2978;Isochinolin;Isochinolin [Czech];NSC 3395;UNII-JGX76Y85M6;beta-Quinoline;
Package: 25kg�����,50kg 200kg
Uses : as an intermediate in organic synthesis and is widely used in multiple fields,
Molecular Structure:
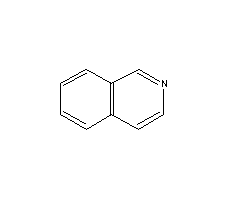
Product description:
What is the chemical of Isoquinoline ?
Appearance: Colorless flaky crystals or liquids to light yellow ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
BP:242-243 °C
===============================================================================
Usage: Isoquinoline is mainly used as an intermediate in organic synthesis and is widely used in multiple fields, including:
Pharmaceutical industry: used for synthesizing drugs (such as antimalarials, praziquantel) and insecticides;
Dyes and color film sensitizers: used as raw materials for producing dyes and color film sensitizers;
Rubber industry: used for manufacturing rubber vulcanization accelerators;
Analytical chemistry: used as a gas chromatography stationary phase and analytical reagent;
Other applications: It can also be used as chemical reagents, spices (such as preparing vanilla essence) and as research and development materials in scientific research.
===========================================================================
Production method: Phenol is mainly produced through the following two methods in industry:
Cumene Process - Currently, over 90% of phenol worldwide is produced using this method.
Step 1: Benzene and propylene undergo alkylation reaction under acidic catalyst (such as phosphoric acid/diatomaceous earth) to produce isopropylbenzene.
Step 2: Isopropyl benzene is oxidized by air to isopropylbenzene hydroperoxide.
Step 3: Isopropyl hydroperoxide undergoes rearrangement and cracking under acidic conditions to produce phenol and acetone (by-products).
This process is efficient and economical, and is the core route of modern chemical engineering.
Sulfonation method (traditional method)
Benzene reacts with concentrated sulfuric acid to produce benzenesulfonic acid, which is then melted with alkali to produce sodium phenolate. After acidification, phenol is obtained.
This method has high energy consumption and pollution, and has been replaced by the isopropylbenzene method, which is only used on a small scale or in special occasions.
