Hydrocinnamaldehyde
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 104-53-0
- EC NO : 203-211-8
- Molecular Formula : C9H10O
- Main Specifications : 99% min
- Synonyms : Hydrocinnamic aldehyde;3-Phenylpropan-1-al;3-Phenylpropanal;Benzyl acetaldehyde;Dihydrocinnamic aldehyde;beta-Phenylpropionaldehyde;Hydrocinnamaldehyde;Hydrocinnamide;Phenylpropyl aldehyde;
Package: 25kg,50kg 200kg
Uses : used in the field of essence
Molecular Structure:
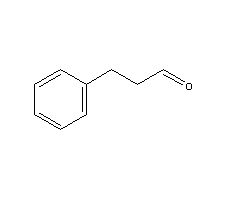
Product description:
What is the chemical of Hydrocinnamaldehyde ?
Appearance: Colorless to pale yellow liquid. It has a fragrance similar to hyacinth, with a burnt sweet green aroma and warm sweet spicy aroma, with a strong and long-lasting aroma ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: At lower pressures (such as 12 mmHg), the boiling point is 97-98 ° C;
=============================================================================
Use: Phenylpropanal is mainly used in the field of essence, perfume and food additives.
Essence and spice: It is widely used to prepare various floral essence, especially lilac, jasmine and rose essence. It can also be used for hyacinth, carnation, lily of the valley and other fragrance types, with the effect of sweetening and enhancing the aroma.
Food additive: According to the provisions of GB 2760-1996, phenylpropanal is a permitted food flavor, which is mainly used to prepare grape, cherry, peach, almond, spice and other essence
============================================================================
There are several methods for industrial preparation of benzal-dehyde, including the following:
Method 1: Cinnamaldehyde is used as the raw material and synthesized through catalytic hydrogenation reaction. This is a common and direct synthetic pathway.
Method 2: Synthesis is carried out using phenylpropanonitrile as the starting material.
Method 3: Prepared by hydrolysis and other reactions of cinnamaldehyde diethyl acetal
