Diphenyl ether
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 101-84-8
- EC NO : 202-981-2
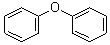
- Molecular Formula : C12H10O
- Main Specifications : 99% min
- Synonyms : 1,1'-oxybisbenzene;diphenyl ether;Diphenyl Oxide;phenoxybenzene;1,1'-oxydibenzene;Dipnenyl ether;Di(diiphenyl) ether;Biphenyl oxide;
Package: 25kg 50kg drum or bag
Uses : As an important component of organic high-temperature heat carriers
Molecular Structure:
Product description:
What is the chemical of Diphenyl ether?
Appearance: Colorless oily liquid or crystal, with a light and clear aroma of geranium, sometimes with the scent of eucalyptus oil or chrysanthemum ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 257–259℃;
========================================================================
Application: Diphenyl ether has a wide range of uses, mainly including:
High temperature heat transfer medium: As an important component of organic high-temperature heat carriers, it can be mixed with biphenyl in proportion to form "conductive A" (with a usage temperature of up to 399 ℃), or with methylbiphenyl to form "conductive LF" (with a solidification point as low as -30 ℃, suitable for low-temperature environments).
Spice industry: It is used as food flavor and daily chemical essence, and can be used to prepare rose, fragrant leaf, hyacinth and other scents. It is commonly used in soap, detergent, cosmetics and citrus preservation.
Organic synthesis: used as an intermediate in the production of flame retardants (such as decabromodiphenyl ether), dyes, and other fine chemicals.
Industrial solvent: used as a stationary phase in gas chromatography, with acetone as the solvent.
============================================================
Synthesis route: Diphenyl ether is mainly prepared through the following two industrial methods:
Reaction of potassium phenolate with chlorobenzene: In the presence of copper powder catalyst, potassium phenolate reacts with chlorobenzene under heating and pressure, and the product is obtained by cooling, separation, and vacuum distillation.
Condensation of chlorobenzene and phenol: In a caustic alkali solution, copper is used as a catalyst to carry out a condensation reaction between chlorobenzene and phenol (molar ratio of approximately 1.06:1.4). After the reaction, acid treatment, layering, and vacuum distillation are carried out to obtain the final product
