4-tert-Butylcatechol
-
- Category :
Intermediates/Pharmaceutical intermediates
- CAS NO : 98-29-3
- EC NO : 202-653-9
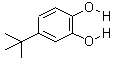
- Molecular Formula : C10H14O2
- Main Specifications : 99% min
- Synonyms : 4-(1,1-Dimethylethyl)-1,2-benzenediol;4-tert-Butylpyrocatechol;4-tert-Butylcatechol solution;Tris(2,3-Dibromopropyl-1)Isocyanurate;Butylcatechol;4-tert-Butyl-1,2-dihydroxybenzene;4-tert-Butyl Pyrocatechol;4-tert-butylbenzene-1,2-diol;p-tert-Butylcatechol;TBC;
Package: 25kg 50kg drum or bag
Uses : used in the distillation or storage process of olefin monomers
Molecular Structure:
Product description:
What is the chemical of 4-tert-Butylcatechol ?
Appearance: Colorless or white to light yellow crystal flake or block form, and can easily become a viscous liquid solution after absorbing moisture ;
Assay: 99% min by HPLC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: 52-55 °C;
===============================================================================
Its main uses include:
Efficient polymerization inhibitor: used in the distillation or storage process of olefin monomers (such as styrene, butadiene, chloroprene, etc.) to prevent polymerization reactions.
Antioxidants: Used in polymers (such as polyethylene, polypropylene, nylon, synthetic rubber) and compounds such as oils, lubricants, and ethyl cellulose to delay oxidative degradation.
Stabilizers and passivators: Used as passivators for amino ester catalysts, as well as stabilizers for insecticides and organic compounds
==============================================================================
The commonly used method in industry is the catalytic condensation reaction of catechol with tert butanol or isobutene. Specifically, catechol and tert butanol undergo condensation in the presence of xylene solvent and phosphoric acid catalyst. After the reaction, the product is purified by sedimentation separation, neutralization, water washing, vacuum distillation, and petroleum ether recrystallization; Another route is the direct condensation of catechol and isobutene in the presence of acidic catalysts such as phosphoric acid or sulfuric acid, with similar subsequent processing steps.
In addition, laboratory scale synthesis can be achieved through Friedel Crafts alkylation reaction. For example, using benzo [b] 1,4-dioxane as the substrate and anhydrous aluminum trichloride as the catalyst, heating and refluxing in benzene solvent, after the reaction is completed, p-tert-butylcatechol is obtained by quenching with saturated ammonium chloride solution, extraction with ethyl acetate, and purification by column chromatography. one
In recent years, green synthesis methods have also received attention. For example, the reaction of catechol with methyl tert butyl ether under specific catalysts (such as NKC-9) produces methanol as a byproduct, which can improve product purity and reduce wastewater pollution.
