2-Hydroxy acetophenone
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 118-93-4
- EC NO : 204-288-0
- Molecular Formula : C8H8O2
- Main Specifications : 99% min
- Synonyms : 1-(2-Hydroxyphenyl)ethanone;o-Acetyl phenol;1-(3-hydroxyphenyl)ethanone;2-Hydroxyphenyl methyl ketone;FEMA 3548;AKOS 90575;AKOS BBS-00003239;2-HYDROXYPHENYL METHYL KETONE;2-ACETOPHENOL;O-HYDROXYACETOPHENONE;O-ACETOPHENOL;2'-Hydroxy acetophenone;2-Hydroxyacetophenone;2-Acetylphenol;Ortho-Hydroxyacetophenone;
Package: 25kg�,50kg 200kg
Uses : naturally present in alcoholic beverages
Molecular Structure:
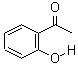
Product description:
What is the chemical of 2-Hydroxy acetophenone ?
Appearance: Light yellow to brown transparent liquid ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
boiling point: 213 ° C (at 717 mm Hg pressure);
==============================================================================
Purpose:
Spice additives: naturally present in alcoholic beverages (such as rum, whiskey), coffee, cocoa, tomatoes, stir fried beef, etc., used as seasoning ingredients to give sweet, phenolic, honey or tobacco like aromas.
Pharmaceutical intermediate: It is an important intermediate for the synthesis of the antiarrhythmic drug "cardiolipin" (propafenone).
Organic synthesis: used for synthesizing other chemicals, such as preparing 4,2 '- dihydroxychalcone through aldol condensation reaction.
==============================================================================
Synthesis route: 2 '- Hydroxyacetophenone is usually synthesized through Friedel Crafts acylation reaction. The specific method is to dissolve phenol in an organic solvent inert to the Friedel Crafts reaction, add anhydrous aluminum trichloride (AlCl3, molar ratio 3-4) in batches, and add acetic anhydride (acetic anhydride) dropwise under heating and stirring conditions. After the reaction is complete, the product can be obtained by hydrolysis with ice hydrochloric acid, liquid separation, and vacuum distillation. The reaction mainly generates ortho isomers (about 80.5%) and a small amount of para isomers, with a total yield of 90.2%.
