1-Methylnaphthalene
-
- Category :
Organic chemicals and Derivatives/Aroma compounds
- CAS NO : 90-12-0
- EC NO : 201-966-8
- Molecular Formula : C11H10
- Main Specifications : 99% min
- Synonyms : 1-methylnaphthalene (alpha);α-Methylnaphthalene;
Package: 25kg,50kg 200kg
Uses : a wide range of applications in industry and scientific research
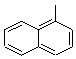
Molecular Structure:
Product description:
What is the chemical of 1-Methylnaphthalene?
Appearance: Colorless to pale yellow oily liquid with a naphthalene like odor ;
Assay:99%min by GC ;
IR Identity: conform to standard ;
HNMR: conform to standard ;
carbon spectrum: conform to standard ;
Water by K. F.:0.5% max or as per the customer’s request ;
Loss on drying:0.5% max. or as per the customer’s request ;
Melting point: -22 °C;
boiling point: 240-245 ° C (atmospheric pressure);
==============================================================================
Usage: This compound has a wide range of applications in industry and scientific research, mainly including:
As a printing and dyeing carrier, it is used in the dyeing process of polyvinyl chloride (PVC) fibers and polyester to help the dye distribute evenly.
Used as a solvent and heat carrier, suitable for high boiling point reaction systems and industrial heat transfer media.
As a raw material for plasticizers, fiber dyeing aids, and surfactants.
Used as a sulfur extractant, pesticide emulsifier (such as 666), and diesel cetane number tester.
As a pharmaceutical intermediate in organic synthesis, it is used to produce fluorescent whitening agents, 1,4-naphthalenedioic acid, naphthylacetic acid, etc.
It is also used in the field of essence and fragrance.
==============================================================================
synthetic route
The synthesis of 1-methylnaphthalene mainly occurs through the following pathways:
Separation from coal tar: In industry, 1-methylnaphthalene is one of the by-products of coal tar distillation, which is separated and purified through methods such as distillation and crystallization.
Chemical synthesis: It can be prepared through routes such as the condensation reaction of methylated naphthalene or o-xylene with styrene. For example, there are literature reports on the synthesis of derivatives such as 1,4-naphthalenedioic acid through oxidation reactions using 1-methylnaphthalene as the raw material.
Other methods include using catalysts for alkylation reactions, or synthesizing naphthalene derivatives by reacting organic metal reagents (such as Grignard reagents).
